Of Microscopes and Myopic Hypotheses
Surely somewhere somehow in your life you've used a microscope. If this was in grade school, perhaps the teacher set it up for you, but most do not escape high school or college, even as non-science types, without using one at some point. Here is your basic microscope you might encounter in a biology or forensic chemistry lab or such.
 The light shines up from the bottom, through your sample, up through the objective lens that magnifies the image and through the eye tube to your eye. The eyepiece usually adds additional magnification (10X). A choice of three objective lenses that can be "dialed in" is quite common. Note the different lengths of these. The shortest lens is the lowest magnification lens and is often called the low power objective. As lens length increases so does the magnifying power of the lens. The technique for using the microscope is pretty universal and begins with something that sounds rather silly: Finding your sample when you look through your microscope! If there's dust on the lens or the stage, etc., depending on what you're looking for, you might find yourself looking at something other than your sample. Dumb as that sounds, it's far more common than you might think, especially if what you're looking at is a hair or a fiber to begin with! The focus knobs work to adjust the vertical height between the sample and the objective lens -- this is called the working distance -- within the range of heights you see your sample, outside that range you basically see nothing. The working distance is the longest for the low power objective and can be very small indeed for the higher power objective (which, incidentally, tends to be the most expensive and delicate of the objectives)
The light shines up from the bottom, through your sample, up through the objective lens that magnifies the image and through the eye tube to your eye. The eyepiece usually adds additional magnification (10X). A choice of three objective lenses that can be "dialed in" is quite common. Note the different lengths of these. The shortest lens is the lowest magnification lens and is often called the low power objective. As lens length increases so does the magnifying power of the lens. The technique for using the microscope is pretty universal and begins with something that sounds rather silly: Finding your sample when you look through your microscope! If there's dust on the lens or the stage, etc., depending on what you're looking for, you might find yourself looking at something other than your sample. Dumb as that sounds, it's far more common than you might think, especially if what you're looking at is a hair or a fiber to begin with! The focus knobs work to adjust the vertical height between the sample and the objective lens -- this is called the working distance -- within the range of heights you see your sample, outside that range you basically see nothing. The working distance is the longest for the low power objective and can be very small indeed for the higher power objective (which, incidentally, tends to be the most expensive and delicate of the objectives)And so, good practice is always to use the lowest power objective first. For one thing it will allow you to "find" you sample most rapidly, but it will also reduce the risk of damaging/contaminating the lens and/or your sample by bottoming the lens out onto your sample. The lowest magnification is also the most forgiving one. Unless your microscope is totally out of whack from the last user, your chances of seeing at least something of your sample on first peek is generally pretty good. But if not, you would use the coarse focus knob and without too much trouble you locate your sample and adjust the fine focus to see a clear image. At this point you are looking at a circular area of your sample. This may appear to be a large circle to you, but the actual area on the sample will be rather small. We call this the field of view. You are only viewing a small portion of your sample, even at this low magnification. Generally the image at this point is of insufficient magnification to see the detail you are ultimately after. So, now you would rotate the next higher power objective into place, then you may require a little coarse adjust, but the fine focus might do the trick to clarify this more magnified image. When you increase the magnification you are decreasing, dramatically, the size of your field of view. And if you proceed to the highest power objective the field of view gets even smaller still.
Now, let's say you have a smear of cells from a biopsy and you get cocky and decide you're just going to get the thing into focus at the diagnostic magnification (whatever is required to distinguish cancer cells). You are looking at a teeny part of the whole sample. Even if you scan back and forth and side to side (more advanced microscopes have knobs that move the stage in these directions for just this purpose), you are so focused in you can easily miss something. If you're following proper procedures, on the other hand, you get the sample into focus on low mag and scan the full sample to look for areas of interest. Once the entire sample is assessed for "areas of interest", you zoom in on each one to see what's really going on there.
Hence both the low power and the eventual diagnostic power objectives serve a purpose. The low power objective is good for seeing the full picture -- not missing any cancer cells in our analogy -- but it is useless for anything more than identifying where there might be a problem. The diagnostic (higher) power objective is instrumental for viewing a small area in detail sufficient to find tell-tale signs of cancer cells, but it would be rather useless for scanning the entire sample, and you would be far more likely to miss something by trying.
Where am I going with this? Well, to this whole discussion of "fat tissue regulation". Gary Taubes wants to look at a close-up of fat tissue metabolism -- that is only at the cycling of the triglyceride/fatty acid cycle within or in and out of the fat cell -- and extrapolate the regulatory events in play there to be representative of overall regulation of fat tissue in the body. So far, he's gotten away with it in certain circles. But Taubes is using a high power lens, and thus he doesn't see what goes on elsewhere, and he doesn't want you to see it either. To do so would mean having to discuss what is out of his myopic field of view. Now it's nice to know the biochemistry of the TAG/FA cycle and how it is regulated within the fat cells themselves, but this is not by any means the right way to go about looking at the overall regulation of fat tissue mass in the body as a whole. For that we need the low power lens to find the "interesting" phenomena going on throughout the body in conjunction with several high power images.
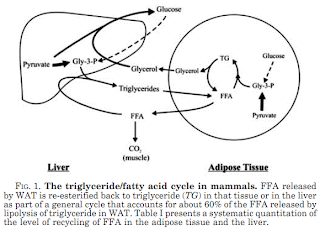
Just a small case in point, let's look at the diagram on p. 149 of my ebook version of WWGF:
And let's compare that even to the depiction in the 2003 Reshef et.al. article referenced in GCBC.
 So the image in WWGF ignores the liver. What else does this ignore? Dietary triglycerides. The relatively huge loads delivered by chylomicrons a couple hours or so after a meal -- whether or not carbohydrates are ingested. Look at the diagram at right. It's a bit blurry, but the green in the bars on the bottom is triglyceride content of the various lipoprotein particles. Chylomicrons have a higher percent triglyceride content. The balls indicate the relative sizes of lipoproteins. The chylo is more than 3X the diameter of a VLDL particle, the main conveyor of triglycerides produced in the liver. The volume of a sphere is V = (1/6)πd3. Therefore a typical chylo delivers over 30X the fatty acids in the form of triglycerides to the adipose tissue if the trig content by percent is equivalent for all particles. But the delivery load is even greater still given the higher proportion of triglycerides in chylo vs. VLDL.
So the image in WWGF ignores the liver. What else does this ignore? Dietary triglycerides. The relatively huge loads delivered by chylomicrons a couple hours or so after a meal -- whether or not carbohydrates are ingested. Look at the diagram at right. It's a bit blurry, but the green in the bars on the bottom is triglyceride content of the various lipoprotein particles. Chylomicrons have a higher percent triglyceride content. The balls indicate the relative sizes of lipoproteins. The chylo is more than 3X the diameter of a VLDL particle, the main conveyor of triglycerides produced in the liver. The volume of a sphere is V = (1/6)πd3. Therefore a typical chylo delivers over 30X the fatty acids in the form of triglycerides to the adipose tissue if the trig content by percent is equivalent for all particles. But the delivery load is even greater still given the higher proportion of triglycerides in chylo vs. VLDL.In WWGF, Taubes reiterates a common statement of his:
... anything that works to promote the flow of fatty acids into your fat cells, where they can be bundled together into triglycerides, works to store fat, to make you fatter. Anything that works to break down those triglycerides into their component fatty acids so that the fatty acids can escape from the fat cells works to make you leaner.
He wants you to look through his "cool and learned" high power objective lens to see the truth, and yet he focuses his microscope on only the part he wants you to see. But it is interesting that we are guided to stop thinking (rightly) of the fat cells as passive depots for fatty acids, but rather as metabolically active entities. And yet, we are equally encouraged to largely ignore the all of the hormones (of which leptin and ASP are but two) secreted by the adipocytes themselves. The very active behavior observed that has elevated adipose tissue the level of endocrine organ.
There is little doubt that Taubes has the part about the triglyceride/fatty acid cycle at the zoomed in level of the fat cell correct. Insulin is the primary regulator of this continual cycle and the balance of it in the post-absorptive state -- that is some hours after a meal through the fasted state until the next one. At that time, the balance is regulated by basal insulin levels to ensure adequate fatty acid supplies to meet energy needs. But this is the big picture of a small part of whole picture. But its like focusing in on a high power image of a biopsy tissue sample, seeing no cancer cells and declaring an "all clear". How about that cell over there, out of the field of view?
In my next post I'll address the whole picture. The regulation of total fat tissue levels is a much more complicated thing than the myopic focus on just the triglyceride/fatty acid cycle.

Comments
Yes, his theories might be wrong in more ways than you'll have time to show us before you retire, but I'll come out in his defense. His book is the only cogent framework on diet that I have been able to follow successfully. I personally didn't -- and still don't -- care whether it's insulin, schminsulin, peptins, leptins, muons or unicorn pixie dust. I lost 30# and got off my blood-pressure meds and statin simply by cutting refined sugar, bread, potatoes, rice and pasta from my diet. I'm not interested nor seem to need to eat at zero- or near-zero-carbs. I eat plenty of animal protein, but don't eat butter until it drips down my arms. And I don't skimp on fruit, unlike his recommendation. But Taubes' general framework is good for me.
So, here I have your blog: had I tried to use it to get healthy, it would have been totally useless. In fact, you are so obsessed with repeating that Taubes is an idiot, that you offer no actionable point of view. Key word is "actionable".
Taubes drew a hypothesis which is likely wrong in many places, but which in my opinion and that of many others is far superior than the ultimate guide to the wrong advice: the USDA's Food Plate (nee Pyramid).
If you want to help the world, perhaps you can spend some of your intelligence and knowledge to discredit the worst offender: the low-fat theories.
Thanks for your advice on what I should blog on, I'll keep my blog title and focus dispelling low carb lies.
Atkins has been around almost 4 decades now Richard. So Taubes' dietary framework is nothing new. But the "fad" part of it is alive and well.
Addendum: Your overuse of acronyms has a serious negative effect on the readability of your posts.
Maybe a mnemonic-type devise can help here? When I see Taubes Wrong Insulin-Carbohydrate Hypothesis Of Obesity I think to myself, "oh, that's both Guyenet's and Evelyn's Struggle to Unravel the Non-relevant Diet Hypothesis Espoused Incessantly by Taubes" and it all makes sense.
When CS posts TWICHOO, I think GESUNDHEIT!
Hope this helps. (-:
I've felt as you do in the past. If it works who cares why. I've since reversed that position as I look at other factors in the broader picture a lot of which I would never have even heard of if it hadn't been for THIS blog whose purpose isn't to tell me how to eat but to debunk food "rules" based on theories that are incorrect and even misleading whether by deliberation or not.
I was never obese and I was never buying low fat cookies. I was never an overweight vegetarian living on frozen tofurkey lasagna and soy ice cream and wondering why I wasn't the picture of health [example of another false belief...animal foods are bad for you and make you sick].
I was never a low income atkins hopefull bypassing the potatos and bananas and beans and oatmeal to buy frozen ground beef patties, bacon, and heavy cream to live on and expecting good results.
What would change for that person if they knew the insulin hypothesis was garbage?
People have been told in general terms what "food" looks like. It probably doesn't come from a box with a cartoon on it and it will have an expiration date in the near future.
They balk. Take away the sugar and fat [and seasonings] in large proportions and they balk. Tubers and fruits are pretty comparable in terms of both carbs and calories until you doctor up the tubers. Not only do people not overeat potatos served plain they express disgust. I have never witnessed anyone eat six bananas in a row [but I could do it with cookies!]. People dislike whole grains compared to refined grains and they like them even better if they are spongey breads with nice fatty toppings or served as a side to a fatty dish.
Even the refinement of processed cereals makes a difference. Rice crispies has a cal count of 130 for a 1.25 cup serving. Pour that into a bowl and acknowledge that you'll be eating at least twice that serving to be satiated and that will include quite a lot of milk and probably a spoonfull of sugar. Rolled oats on the other hand are 150 cals for a single 1/2 C dry measure serving which will fill me personally through the entire morning.
So if I choose rice crispies I'm eating 300 cals easy and probably more. If I choose oatmeal I'm eating 150 to feel the same degree of satiated if not moreso. Apply that to every meal every day and it makes a difference. It is actually rather difficult to exceed cal needs on a high carb whole foods diet, the more mixed it is the easier it becomes.
I suspect the fact that people are looking for a "program" or a "system" [or even a blog] to tell them how to live and what they are allowed to eat hits closer to the truth of the issue than we formally acknowledge.
It pains me to admit this - but you can be both an idiot and a good writer.
Equally, the low-fat diet has worked for many people (including me) so try not to extrapolate your n=1 evidence to decide what must work best for the whole human race.
I have to say this is the first site I check each day as I find Evelyn's posts so informative and written in a funny, relaxed style so I don't understand your problem on that score either.
@euler: And Obama is (supposedly) a great speaker too ... Sorry to go there gang!
1. Are you at your goal weight/body composition?
2. If not, how do you plan to get there?
3. Do you think obesity is a possible outcome when on a VLCD?
4. Can you point to a single population that is obese that does not eat refined carbs and/or eats the majority of their calories from fats and proteins?
Fred:
1&2: Not relevant to the science, I've shared that info here, but you really might not want to go there ...
3: blogger beware
4: majority is >50% right? SAD anyone?
I know that Fred Hahn's questions weren't directed to me but I can't resist the fun. So pardon me if I butt in.
"1. Are you at your goal weight/body composition?
ANSWER: I am 7 pounds away, closer than I have ever been, as a result of calorie counting and increasing my exercise levels. (No weight lifting, body exercises, walking and hiking.)
2. If not, how do you plan to get there?
ANSWER: By doing what I've been doing.
3. Do you think obesity is a possible outcome when on a VLCD?
ANSWER: Yes.
4. Can you point to a single population that is obese that does not eat refined carbs and/or eats the majority of their calories from fats and proteins?"
ANSWER: I don't understand a convoluted question like that. The overwhelming majority of humankind eats carbohydrate-based diets and until recently experienced no obesity issues. (Asia, Africa, North and South America, I think that about takes in the majority of humanity.)
If Hahn is saying that REFINED carbs are implicated in the obesity epidemic, I'd agree. But somehow I don't think that's what he's saying.
BTW, when I was a Low Carb fanatic I tried his stupid slow burn lifting technique and I think it is one of the reasons I tore a rotator cuff.
ANSWER: I don't understand a convoluted question like that. The overwhelming majority of humankind eats carbohydrate-based diets and until recently experienced no obesity issues. (Asia, Africa, North and South America, I think that about takes in the majority of humanity.)
Thank you for articulating that so well, the framing of the question was leaving me befuddled as well.
_____________________
5. Can you do any more than you already have to become the worldwide poster boy for confirmation bias
6. could you possibly stick your fingers in your ears harder (than they already are) and shout louder (than you already do)
"la la la la la I can't hear you"
"la la la la la I can't hear you"
"la la la la la I can't hear you"
I should have written that I don't do weight lifting. My exercise regimen is based on walking and hiking, with some bodyweight thrown in. I've ordered a 4.5 pound medicine ball.
If you tore a rotator cuff, you weren't doing stupid slow burn. You were just doing something stupid. And i'm sorry you got hurt. ]
However, if you were actually doing Slow Burn correctly and you tore a rotator cuff, you should go see a doctor since you are probably nutrient deficient. A common outcome of lowered calorie diets high in grain-based carbs.
And speaking of "stupid," if you couldn't understand my question, well, I can't help you there.
Since you are calorie counting, how many calories in carbs/sugar minus the fiber are you taking in on a daily basis? And I do indeed implicate refined sugars/carbs as the main culprit in obesity.
And by not weight lifting, I am sure you lost a good deal of lean mass in your weight loss journey. That's to your detriment. Walking does not cause positive tissue remodeling. And exercise hasn't a thing to do with weight loss. Read the research.
Evelyn - I think this blog as well as virtually all your others on low carb diets as bogus are a way for you to justify eating the carbs you love. I believe you still struggle with your weight (I could be wrong but I don't think so) and this is because you won't give up your beloved leaven.
I say this because the only way you are going to lose fat is if you cut the refined/total carbs as Diana did without realizing it. She thinks it the calories but the body doesn't have calorie receptors.
Sanjeev - That was a very intelligent response. You really got me there.
Bentley said:
"The overwhelming majority of humankind eats carbohydrate-based diets..."
References please.
Is this a common problem for you? Might explain a lot.
"The overwhelming majority of humankind eats carbohydrate-based diets..."
References please."
No, it was Diana who said that.
Doesn't everyone just love it when people who can barely read think adolescent cracks about others' intellect are funny?
Oh dear, asking for references.... I realize this is hopeless, but:
China
Japan
India
The Pima Indians.....
Indeed, if you are looking for what to eat, it's pretty damn simple, and minus the high reliance on cereals, the food pyramid is not that bad, actually. Lots of veggies and non-processed food, control your portion size, have a treat here and there, and that's it! No need for a blog or a book for that.
@Fred
Are you blaiming carbs, or refined carbs? Because there's an whole world between both. I don't think refined carbs per se are responsible either anyway, as clearly it's all about energy expenditure and getting calories from sugar or potatoes won't do squat as regard to body composition (see this http://www.ncbi.nlm.nih.gov/pubmed/9094871)
but i'm still curious why one would talk about carbs and refined carbs as if they are the same.
As for references that most human thrive well on high-carb diet, Katharine Milton gives about the best perspective on HGs so far in the litterature (see http://www.ncbi.nlm.nih.gov/pubmed/10702155) and even if you go with Cordains or Eaton, you will see figure like 60-70% plants, 40-30% animal derived foods. There's a lot of evidences about Okinawans and Kativans & other that did very well on a very high-carb diet.
2 mins on google scholar and you can find about anything.
@Diana
You should find a knowledgeable coach and start doing heavy resistance training. Nothing work best with a caloric deficit + slightly higher protein diet for body recomp. :)
The Pima Indians ...
One little two little three little Pimas
Four little five little six little Pimas
Seven little eight little nine little Pimas,
Ten sprightly carb-eating Pima boys (and girls)
... and no acronyms!
I'm reading a book about Yoga right now, which claims it is a recent Western creation and not an ancient Indian practice. (Sorry Sanjeev!). The reviews are uniformly excellent. In brief, they admit that what the author says is true, but they "still believe" that Yoga is something it's not.
(Book is THE YOGA BODY by Mark Singleton.)
Going very low-fat was probably a bad idead but it's neither better to go crazy about fat. Fatty acids can be nasty metabollicaly speaking, just as can be glucose and protein (they might be the worst actually). Everything should be reduced, and this is why calorie restriction is the only possible mean of reducing food-induced oxidative stress/inflammation and possibly expend lifespan (also by other means than reduced excessive oxydative stress/inflammation).
That's something that never really gets talk about actually but that's something that i'm always wondering. If CICO is invalid, why do calorie restriction constantly works? How can we be sure to be calorie restricted if calorie are bullshit and the human body is too complex for that?
SPRIGHTLY AND HEALTHY.
Let us all repeat now: SPRIGHTLY AND HEALTHY. 100 reps!
@Frank, I have started resistance training, esp. upper body. I just wanted to get my eating habits in shape first. First things first. Thanks for the tip though. And I am looking up that study you cited.
Having said that I still have to gasp when anyone demands a "reference" for thin carb-eating cultures - if they can't open up their lying goddamn eyes, how many studies will convince them?
Sounds too much like exercise ;)
that's rich.
Coming at the end of a series of posts full of unreferenced, unproven BS claims; ASSertions provided with ZERO proof ...
You again fulfilled one of my predictions (predicted on this blog before): demand voluminous proof from others that you never willingly provide yourself.
> Sanjeev - That was a very intelligent response. You really got me there.
I don't need to "get you" FRED. Your antics let people "get you" all on their own, on a gut level.
I was just making the gut level understanding explicit; lots of folks still don't know what confirmation bias is, maybe I'll spur them to look it up, and line up the definition with your behaviour.
PS - You should hang out on homeopathy, UFO, bigfoot, 9/11 conspiracy & anti-vaccination boards - great place to find others with similar mental patterns. Just trying to help you find a social / intellectual milieu that suits you.
Oh man, that's why we need CARBSANE!
A friend who does yoga told me she's never seen a place in North America that teaches the meditations that are supposed to come before the physical practices.
Wouldn't surprise me at all. Like some versions of "ancient" kung fu taught here but don't exist anywhere else ... I know zip about Yoga myself, except as a place to meet some of those ballerinas we talked about earlier
I'll have to find it at my library, thanks for the tip
If I am recalling correctly from her previous blog posts, her point is that she lost quite a bit of weight on LC/VLC and then hit the dreaded sticking point that many low-carbers hit. Instead of giving up and blaming a damaged metabolism, she's trying to learn more about diets and health in the hopes of finding what works. That seems, to me, to be the ideal approach to becoming leaner and healthier.
I don't know if it's possible to become obese on a low-carb diet. My inclination is to think that it's very difficult, perhaps impossible. But that point only helps the 350 pound person who is looking to lose large amounts of excess fat. It doesn't help the 180-250 pound person who wants to lose those last 20-80 pounds and finds that ad libitum LC is no longer living up to the promise, and refuses to accept that he is at some ideal weight or suffering from a busted physiology.
I like this site because it's a place to come to when you finally accept that there is no magic pill that will solve our health and weight issues. This is where we come when we're ready to roll up our sleeves and get some work done.
The question is, what is it about it that escapes you? Be honest. I have dealt with FAR too many clients to be fooled by your rhetoric.
IOW, you cheated. You indulged. Sure you did. You went face down in the za or bagels or whatever starch or grains that gave you pleasure. Sure you did - just like everyone else who loses and then experiences no more loss.
You are saying that all stalls on VLC are because of cheating?!!
Fred said “You went face down in the za or bagels or whatever starch or grains that gave you pleasure”
You do realize that you just said that Food Reward trumps high-satiety VLC dieting…
I haven't gone face down in anything in over five years. You on the other hand snarfed down five lobster tails no doubt drenched in butter in one sitting. Great example there man!
OK, Carnac the Magnificent (google this if you don't know who that is): what did I have for lunch?
(Analyze This hilarious scene)
My favorite was Aunt Blabby.
@Evelyn,
How funny, in the last 5 months of 20 pound weight loss, I've eaten bagels, rice, etc., all the neolithic agents of disease (except pasta). On the other hand, I went up to 156 pounds while stuffing my pie-hole with gargantuan meat and salad meals.
Wonder how that happened? Hm. Back to the drawing board.
(HOWSOME-EVER....cheating is a big problem on Low Carb. Most people end up binging on carbs at some point. The whole "forbidden fruit" thing.)
> that Food Reward trumps high-satiety
> VLC dieting…
yeah staying on that low carb diet is easier than falling off a floating log
heck, it's SO UNBELIEVABLY EASY, many who have financial incentive, who've staked a career on it can NOT stay on it ...
<snicker> oh, wait ... </snicker>
> loss, I've eaten bagels, rice,
> etc., all the neolithic agents
come clean Diana ... they were low carb, low fructose, low PUFA, gluten free, free range, grass-fed bagels.
And the only way you did it was insulin injections directly into the hypothalamus
$&^%&*&^(^$(#*$(#*^$(#$(*#^$(.....
%A*&#$*#&$*#&$%*A&%*@%#@(*)(#*@_*($&#...
>>>>>>>>............
IF YOOUUU DON'T EAT LOW CARB YOU WILL DIE!!!! (soon)
I dunno folks, maybe we should give Fred a break. He can sure be entertaining at times, and after all, he did "discover" me by emailing Taubes about this blogger he needed to silence ;)
Here's the ultimate irony. I can understand the LC fervor, not only because I've been there but...because I'm still there.
For me, and I suspect a lot of people, the macronutrient that will give you the most problems is carbs. If you want to get a handle on your calorie intake you will have to zero in on those delicious pesky carbs. For a lot of people simply cutting their carb gram intake in half might do the trick. That DOESN'T mean LC is right.
"gluten-free" - I don't understand the fuss. Isn't gluten a protein? Doesn't that officially make it "good"?
When dogmas collide.....
OK I admit it.
Grass-fed? Bah humbug. I actually ate grass.
Yes, I snuck into Central Park after hours (the only place in NYC where there IS grass), and chowed down.
I brought my homemade perfectly balanced O3/O6 dressing with me.
:):):)
Fred: "I have dealt with FAR too many clients to be fooled by your rhetoric."
In other words, empirical evidence trumps n=1. :)
> Let's run Fred's comments through the
> Universal Translator (from Star Trek):
Babel fish would be better but those are way too low fat for FRED's tastes[0] .
http://en.wikipedia.org/wiki/Babel_fish_%28The_Hitchhiker%27s_Guide_to_the_Galaxy%29#Babel_fish
Keep in mind, no translators work where FRED is, trapped in a low-carb Tholian web inside a Dr. Who style time loop[1] circa 2007/8 (when low carbers were multiplying like tribbles)
http://en.wikipedia.org/wiki/The_Tholian_Web
http://tardis.wikia.com/wiki/Time_loop
I'll end here ... no need to bring up the centaurian slugs (used by Nero) or Ceti Eels (from "Star Trek II: The Wrath of Khan") in the brain stem making people believe in low carb
[0] tribbles are high fat, so maybe he prefers eating those
[1] also known as Ground Hog Day, but with zero humour when FRED's involved.
I wonder if FRED will ever tire of being the Tetsuo to Gary's Kaneda.
and
"She thinks it the calories but the body doesn't have calorie receptors."
Why even ask about macronutrient proportions of calories eaten, when calories are irrelevant?
These LCers can't seem to make up their minds about calories!
Do they matter?
Do they exist?
They don't matter and they don't exist but it's important that you get most of what doesn't matter and doesn't exist from fats and proteins.
That makes total nonsense.
That makes total nonsense.
That about sums up Fred. Of course he's always respectful when he accuses folks of being cheating, lying, addicts making excuses for their addictions. But apparently I was snarky for suggesting he turn his psychoanalytical powers towards helping his friends solve their weight issues.
Someone really ought to get these folks on the same page. I could have sworn Andreas was making the "we subbed carbs for fat and that made us fat" argument just a few weeks ago. It's hard to keep up with it all LOL.
And Sanjeev: geek gauntlet! Love that!
Doesn't Quark remind you of anyone? >:)
That's also not to mention a claim he made on his site that animals in the wild don't get cancer, inferring that cancer is a man-made disease. I'm wondering how he explains cancerous bone growths that have been found on dinosaur fossils. Or how he explains the process of evolution, as one of the mechanisms behind evolution (genetic mutation) is the same mechanism behind cancer.
But it's so much fun. It's like getting shot in the ass by your bratty little brother playing with a toy bow-and-arrow set.
> in the wild don't get cancer
some time ago I could NOT convince a couple of relatives not to take
shark cartilage
or copy & paste
http://www.google.com/search?hl=en&client=ubuntu&hs=wIN&channel=fs&q=shark+cancer+&oq=shark+cancer+&aq=f&aqi=g3g-v6g-j1&aql=1&gs_sm=e&gs_upl=13917l13917l0l14964l1l1l0l0l0l0l699l699l5-1l1l0
my memory of his voice is not letting me think of any real person ... I do get a slight impression of Dr. Eades.
Post a Comment
Comment Moderation is ON ... I will NOT be routinely reviewing or publishing comments at this time..