Mechanical Work, ATP, and Macronutrients (and Thermodynamics)
[Blogstress note: this article is not similar to that in AARR recently mentioned here]
In the background here I've been doing a lot of thinking and writing on thermodynamics and how the body uses food to produce internal energy to "do stuff". In the end, we "burn" the macronutrient molecules in our food to release the chemical energy stored in those molecules, and ultimately we use that energy to do "work".
I've said we can mostly ignore entropy in the context of the human body, but in chemical thermodynamics we have the concept of Gibbs free energy: G. Most importantly in chemistry, we are concerned with the change in free energy of reactions:
ΔGrxn = ΔHrxn - TΔSrxn
ΔHrxn = change in enthalpy (energy) ΔSrxn = change in entropy
ΔGrxn = change in free energy
Free energy is the energy available to do work. For example to move things such as the piston in an engine, an electron through a wire, or to move an ion from one side of a cell membrane to another. In the human body, we are ultimately using energy derived from molecules in our food to support life by "powering" other chemical reactions (synthesis, etc.), and things like moving food through our digestive tract, generating nerve impulses, and moving our body parts to sit, stand, walk, run, etc.etc. A sticking point for a lot of people with thermodynamics relates specifically to the context in which most first learn about entropy: the combustion engine. [You are invited to read some of my other posts on thermodynamics here: Of Thermodynamics, Chemistry, Biology and Biochemistry, Of Thermodynamics, Complexity, Closed Systems & Equilibrium and A Fein(man), Fine Mess of Thermodynamics.] In the human body, most of the "work" done directly by the chemical energy is electrochemical in nature. Still, eventually we do somehow translate that into physical movement, aka mechanical work.
Some analogies can be drawn between a combustion engine and our electrochemical "engine", but others cannot. A major distinction is that in the combustion engine, all of the chemical potential energy is released as heat, which creates pressure due to the gasseous products wanting to expand. This pressure (force) acting on the piston moves it, thereby performing mechanical work. In the human engine, the macromolecules are broken down and mostly converge at the point of a molecule called acetyl-CoA after which point the rest of the chemical "workings" of the engine are the same regardless of the original fuel source.
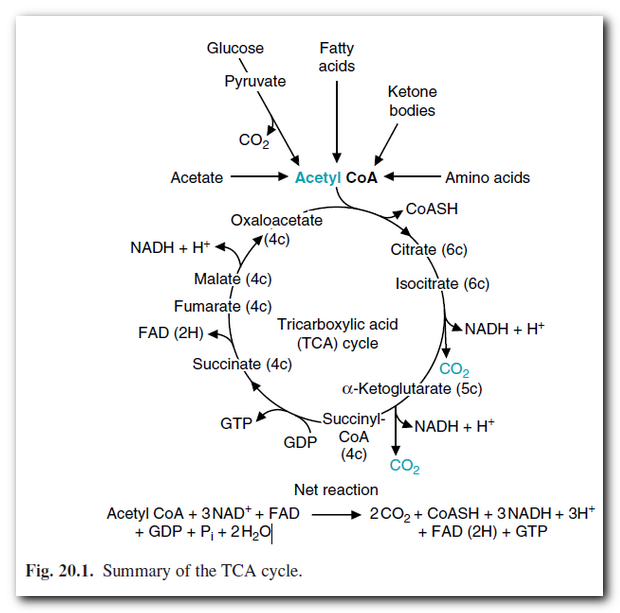
In All Roads Lead Through Krebs, I included the graphic at right from Marks' Basic Medical Biochemistry. This is the Tricarboxylic Acid Cycle or Krebs Cycle, and I've copied it here for reference. Please refer to the post for other ways that some amino acids enter this cycle.
Molecules often referred to as "reducing equivalents" (NADH, FADH2) are generated in this cycle, and these, along with reducing equivalents generated in fatty acid oxidation and glycolysis, are "fed into" what is called the Electron Transport Chain, or ETC. Whatever chemical energy that is lost as heat along the way has already been lost at this point, so we are dealing with a predictable number of these reducing equivalents per acetyl-CoA entering the TCA/Krebs. Unless you are a small animal such as a rodent, the ultimate outcome of the ETC is to use the electrochemical energy to power the phosphorylation of ADP to ATP. In times when heat is needed, this process is diverted or "uncoupled" from ATP generation, but that is a topic for another day.
Moving Off the Grid ~ (analogy time again)
Let's say you want to move away from civilization and rely solely on energy you can harness on your property. You find an ideal location with a strong running stream, frequent bursts of wind, and an open field. Oh ... and you decide that rather than use powered exercise equipment you're going to harness the energy from those as well. Only you hate cardio and learn that Tony Little has moved to the 100 acres next door, so you invite him to come over and use your Gazelle.
All of these are hooked up to a giant battery in your basement and everything in your home is run off of this Main Battery: heat, light, appliances, etc. [Note: this configuration is for the purposes of an analogy only.] So the water wheel basically charges the battery continually, the wind energy in short random intervals and the solar panel in cyclic daily fashion. Tony, well, he just adds a little extra to make him feel good :-) When you turn on your light bulb, it will illuminate if there is enough charge in your Main Battery. Whatever wattage your bulb is will determine how much energy it drains from the battery over the time that it is used. If you are using an inefficient old incandescent bulb to light your room, it drains more of the battery and gives off heat. If you are using an energy efficient LED, it provides the same light while draining less energy and giving off virtually no heat.
The light bulb draws energy from the Main Battery. It needs a certain amount to be illuminated. Your light bulb doesn't care where the energy stored in the battery came from.
The Main Battery provides energy based on the "load" that is placed on it. Your battery doesn't care what it supplies energy for, or how efficiently that device uses the energy.
Adenosine Tri-Phosphate (ATP) ~ Your Main Battery
More than two thirds of all ATP generated is produced through the TCA/Krebs Cycle. The vast majority of cellular activities is powered directly by ATP. It would be fair to say that ATP is the equivalent of the "free energy available to do work". It is also true that all ATP are alike regardless of where they came from. Therefore, from the point of ATP forward, all ATP-powered "work" and the thermodynamics, the energetics, whether or not entropy losses are involved, etc., all of it is the same.
ATP is like the Main Battery in the previous example, only there is no central depot for this internal energy supply, but rather it is contained locally "on site" of every cell. I suppose an analogy could be made for our house that rather than a main battery, there would be some battery network at every socket or directly in the appliances and such. I don't think this is necessary to elaborate, however, in order to draw the following corollaries to the above arguments:
Walking requires ATP to do the mechanical work of muscle contraction. A certain amount of ATP is needed to produce the necessary muscle contractions, etc. The muscle cells involved just use ATP and can't distinguish where it came from.
The amount of ATP required to perform a task is based on the "activity load" (speed, incline, etc.). Each ATP provides the same amount of energy regardless of what activity it is fueling.
So perhaps the water wheel is analogous to fatty acids continually cycling in your blood. And perhaps the windmill is analogous to carbohydrates and the solar panel to amino acids. Tony? Why he's alcohol of course! Pops by to add some energy during the week, perhaps he comes by more than once a day on the weekend. :-)
Dat Entropy ...
I recorded and put three short animations/tutorials available online here (Chapter 10) that shows how muscles contract and the role of ATP. Direct link to my video.
Towards the end of the video when the ATP is attached to the myosin head, is broken down to ADP+P and transfers energy to the myosin head, and the subsequent movement that would be mechanical work is the "equivalent" of the expanding gasses in a combustion engine. How much heat is lost in this process I don't know, but that wouldn't be entropy. Are there entropy considerations so that the full energy stored in the ADP-P bond is not available to do the mechanical work? Probably. Does it matter? Probably not. Not as far as tracing it back to any difference between macronutrient sources. It would be the same for each ATP used in each specific application.
In Summary:
The "Burning:
- All macros are broken down and converge on acetyl-CoA. (Some amino acids feed directly into Krebs at different points)
- The difference along the way is in some direct ATP formation, and the formation of different amounts of different reducing equivalents (NADH, FADH2).
- From acetyl-CoA on down the energy production line through TCA/Krebs and the ETC, all reactions are the same in the production of ATP.
The "Work"
- Once energy is stored in the ATP "batteries", no cell knows the macronutrient origin of that energy.
- All ATP does whatever work -- chemical, electrochemical -- it does through the same reactions and mechanisms.
- Energy use or any losses associated with converting ATP chemical potential energy to other forms is the same regardless of the source of that ATP.


Comments
Clearly this is an error on his part:
The second important point to recall is that ketone bodies bypass this process (i.e., glucose to pyruvate to acetyl CoA), as B-OHB enters the mitochondria, converts into acetoacetate, and enters the Krebs Cycle directly (between succinyl CoA and succinate, for any biochem wonks out there). I keep alluding to this distinction for a reason that will become clear shortly.
Because there's only the "pickup" of CoA from the succinyl-CoA after which it feeds in normally.
That said, any discussion on the energetics of ketones as fuel vs. glucose or fatty acids for that matter is not a fair fight. Ketones are not a primary fuel and there is an energetic cost to create them and convert them back. This may be why exogenous ketones are so powerful -- they are more "speedy" than even straight glucose water! -- but not physiologically relevant.
Most of these neurological disorders seem to have two problems: either GLUT transporter problem or pyruvate dehydrogenase which almost acts like a transporter in the way it recruits pyruvate (hope that makes sense, not the most technically accurate way to describe it).
Speaking of, got this from his website. Bad plastic surgery, bad Photoshop, or both?
Mr. Attia's analysis confused me, I will go over it again, but if he thinks that B-OHB actually enters the Krebs cycle at the Succinyl steps then perhaps I am not as confused as all that. His ETC comments seem to assume that "entry" into the Krebs cycle, otherwise the greater energy differences he talks about make no sense. Or maybe, I just don't see something, that's very possible.
I don't see any special magical qualities in the ketones, no meaningful explication of same, not yet - although, sometimes all these players in our metabolism seem extraordinary.
A few points I didn't pick up:
* Why is the light bulb on in the first place? Maybe you turned it on because you thought Tony Little was coming over. Maybe you turned it on because there was so much electricity coming from the windmill that you figured what the heck. Maybe there was so much sunshine powering the solar panel that you forgot the light bulb was even on.
In any of these cases, "Energy In" and "Energy Out" aren't independent variables. If it weren't for the energy coming in, the light bulb wouldn't be in use in the first place.
* Is there only one bulb? If not, what determines whether the 2nd or 3rd bulb are turned on? Is any of this related to the amount of energy coming in, or to the cost of producing additional energy? If so, then again the Energy In is partially determining the Energy Out.
* Is it possible to upgrade the less efficient bulb for the more efficient bulb? Might there actually be an advantage to having the less efficient bulb? For example, to heat the Easy Bake Oven so you can fix Tony a snack?
A biochemist could tell me that in order to lose 50 pounds of fat, I need to create a caloric deficit of 175,000 calories. But that doesn't actually tell me how to lose 50 pounds of fat. Suppose your accountant told you that in order to become a millionaire, you need to create a monetary surplus of one million dollars. That is undeniably true, yet at the same time doesn't actually tell you how to earn a million dollars.
I suggest to examine the work by Veech RL
The issue of CI-CO not being independent variables has been beaten to death, but doesn't change the fact that CICO holds, nor would it change the point of this post which is that our "internal energy source" is macronutrient agnostic. Of course in nutrition we have certain additional constraints. For example, if birds were to crap all over our solar panel or we had a long span of cloudy days, this would be akin to protein insufficiency. I suppose I could amend my analogy to have solar just power specific essential functions in the home to model this.
I have a question which I am sure has been the subject of some previous posts but I couldn't really find them anymore. I'm not sure it's related to the topic here, but I hope it won't hurt the conversation.
I keep hearing that calorie restriction doesn't work because your body adapts to the lower level of calories and your metabolism slows down which will end up in a rebound once you get back to your normal calorie level.
Now, controlling for the fact that the body that weighs less needs less calories, has there been any study showing whether there is indeed a metabolism adaptation to lower calories that is maintained after returning to your (new) adequate calorie level?
I am asking because I noticed how easily I can drop 5-8 kg in one month by doing a modified version of the 2 day diet with around 700 kcal/day low fat low carb on the restricted days and minor calorie restriction in the rest. I am wondering if this is indeed dangerous as a weight loss strategy and whether returning to my new calculated calorie need would mean gaining weight again.
Thanks!
Daniel
The guy does look pretty damn good (if a little creepy) for his age though.
With the gas station analogy, the gas mileage could be an issue if the nearest gas station is 75 miles in the opposite direction. If so, one would have to drive 350 miles in order to get enough gas to net 200 miles in the direction one wanted to actually travel. Thus, it would take 17.5 gallons to net the 200 miles you actually want to travel. Even though your car is getting 20 miles per gallon, it takes more than 10 gallons to reach a target 200 miles away.
With the light bulb analogy, the light bulb doesn't care whether it is turned on or off. Someone has to make a decision to turn the bulb on. That decision could be very different if the person making it is trying to extend the life of the bulb. Suppose it's the only bulb you will ever own and if it goes out, it's gone forever. Each time you turn on the switch, it's a stress on the bulb. Every hour you burn the bulb brings it closer to the end of its useful life.
You are correct that the bulb doesn't care whether the electricity comes from a nuclear power plant or a hamster wheel, but the question hasn't been answered: "Why is the bulb on?"
re T1 and RQ ie not this post
Is this any help ? http://diabetes.diabetesjournals.org/content/29/5/365
I just found this. Pretty dang impressed. Does he sell out some? For sure. But he's actually more of a real deal than many ... ahem ... "real guys" out there.
http://www.details.com/culture-trends/movies-and-tv/200902/the-infomercial-king-mr-big
>>>Tony Little is little. Not tiny—he's got the pecs pushing against his shirt and the quads that can shove a piano down the hall—but not huge. He wears a cap pulled low, which makes him look younger than his 52 years, and a ponytail of stringy blondish hair. But he doesn't wear fancy clothes or shades. He's polite. He smiles easily. Bottom line: What Tony sells is his story, which sounds both too good and too bad to be true. His dad, an oil-refinery owner whom he calls "an asshole," abandoned him. His mother, a schoolteacher, shipped her unruly teenage son from Fremont, Ohio, to St. Petersburg to live with her brother in 1973. On the Gulf Coast, Tony made some friends in the bodybuilding community, and they helped him become Mr. Florida and a contender for Mr. America. He came within one point of beating eight-time Mr. Olympia Lee Haney. Then came the accident, the day in 1983 when the school bus hit his car and he herniated two disks and wrecked his knee. "I spent two years in and out of hospitals," he says. "I drank half-and-half for breakfast." But wait. There's more. He got a "bad spinal tap," suffered spinal meningitis, temporarily lost his eyesight, spent 18 more days in the hospital, got kicked in the nuts by a horse, spent two weeks on his back, inadvertently sat in some acid, went to a burn center, was hit by a lobster truck, and crashed his car into, he says, "the only hill in Florida," which cost him four reconstructive face surgeries.<<<<
Other studies I've seen have suggested that people who successfully maintain a large weight loss tend to be fairly vigilant about their weight loss maintenance. Most weigh themselves regularly, exercise regularly, and their maintenance diet looks a lot like their weight loss diet, with slightly larger portions (a few hundred calories per day extra than when they were losing).
It's probably a good idea to look at food not just from the calories you are getting, but the nutrients you are providing your body. If you decrease calories coming in, it's a good idea to increase the nutrient density of the foods you are eating.
Swapping out a sandwich and soda for a salad and iced tea isn't just a reduction of calories. You are replacing nutrient poor foods such as white bread and refined sugar for more nutrient-dense foods like green leafy vegetables. From a caloric stand standpoint, you're eating less but from a nutrient standpoint, you're actually eating more.
OTOH, if you normally eat 18 donuts per day and you cut that down to 6 donuts per day, you are not actually getting more nutrients. So, that kind of calorie restriction probably isn't going to be effective long-term.
Other medical conditions aren't treated this way. Show up at the doctor's office with a bullet wound, and they don't say "well, you shouldn't have gotten shot. The number of bullet wounds in your body exceeds the recommendations of the Centers For Disease Control, which is zero. Look at me. I'm twice your age and I've never even been grazed. That's because my healthy lifestyle choices include a low-fat diet, plenty of whole grains, and not getting in the way of flying bullets."
Anyway, I have to go to the gym now....
It's not quite what I'm looking for but informative for what I'm getting at nonetheless. Thank you :-)
I'm not sure if it can be tested what the cause of reduced glucose oxidation is -- insulin deficiency or fatty acids (and/or ketones) "swamping out" the mitos.
Piggy backs on the other paper that demonstrated that the change in REE could account for weight changes after insulin treatment as well. E.g. metabolic cost of gluconeogenesis + peeing out a few hundred calories of glucose.
A red dot was "obese". A green dot was "not obese". She offered no medical treatment to help me obtain this goal. She would not prescribe any pill to curb my appetite. Nor would she consider the possibility that the medications that were prescribed made it more difficult for me to lose weight.
Even after I lost 50 pounds, she would still whip out that stupid height-weight chart because I was still in "obese" territory.
So, "don't be obese". Not in those words, but that was the only medical treatment she offered.
I've seen wehre Veech has been quoted saying if he has a heart attack he wants ketones. Exogenous ketones are not a "fair fight" and their deltaG is meaningless for endogenous ketones as they are an intermediate in the pathway.
The pathways produce the same amount or ATP per molecule so if the differential is probably in heat lost during ATP production. But heat is not "lost" in warm blooded animals.
However, don't you have energy balance no matter what? If the light bulb is off, you still have energy balance, because there is zero energy entering the light bulb and zero energy leaving it.
If the battery is running low, and the bulb is burning a bit dim, you still have energy balance, even though the amount of energy being used is less than normal.
If there's a power surge, and the battery glows more brightly than normal, there's also energy balance.
If you add an additional light bulb, there's energy balance. If you add more batteries, there's energy balance. Energy in and energy out are always the same.
So, if the fact that you have energy balance can't tell you if the bulb is on or off or how much energy the bulb is using, or how much battery capacity you have, or how bright the bulb is, or what kind of fuel you're using, what useful information does "energy balance" convey?
Again, I apologize if I sound like an idiot.
say WHAT ???
Note the "" around lost. In warm blooded animals heat is not waste (as it would be in an engine for example ... please do read the background posts I linked to where I've discussed this before). So if substrate A produces more free energy (that's H) that is "available to do work" but doesn't mean it does work., then some is heat because ATP production takes the same amount of energy from the same intermediary molecules in the same metabolic cycles.
More heat from general metabolism = less need to purposely produce heat.
Endogenous ketones are not a separate entity if that makes sense. If you are going to address some metabolic advantage from ketone forward, you need to address what went to make the ketone. I don't hear a lot about pyruvate or lactic acid. Both are energy substrates but we ingest very little of either.
http://www.dlife.com/diabetes/type-2/diabetes-causes/colberg/causes_of_diabetes?utm_source=Update-20140926&utm_medium=eNewsletter&utm_content=Update-newsletter&utm_campaign=dLife-eNewsletter&
Any differences in oxygen consumption, standardizing ATP to number of carbons or O2 or Delta G to either of those ... all of that has to be different in the steps prior to acetyl-CoA. But even THEN, the ATP produced directly in glycolysis, for example, is the same ATP as what is produced in the mitos originating from a fatty acid.
Each ATP contains the same energy, has the same ability to do work. As you can see with the muscle contractions, this is a stoichiometric release of energy -- 1 ATP, the whole "packet of energy", at a time.
Unlike a car engine, for example, heat that is produced in chemical reactions in warm blooded creatures is not waste. It may not be available to do work, but it reduces the need for uncoupling (robbing energy from ATP production and work) to produce heat purposefully.
So then we get what I used to call the "whyners" because once you demonstrate the obvious we go back to the other canard about how thermo is a tautology and it doesn't tell us anything. Thermo cannot tell us any of these things. That doesn't make it a valid principle.
Gary Taubes and his ilk want people to believe that insulin causes fat to be trapped in the fat cells thereby starving the rest of our cells making us hungry and eat more. But he says the obese don't eat more. Or he said that. I could go on but I can't rehash more today.
You are actually incorrect that we are always in energy balance. We're probably not in energy balance the vast majority of the time, but averaged out over time, a weight stable person is in energy balance. When we eat we gain energy and store it or burn it, when that's gone, we burn the stored energy, and the cycle repeats. Homeostasis is what the body tries to do, but everyone in overfeeding studies gains weight (and fat) and everyone in caloric restriction loses weight (and fat). There has never been a metabolic ward study that has shown otherwise because it is physics and physiology.
Consider this. NuSI is spending almost 14 million dollars to test different diets. Not for weight loss mind you. No, to lose the weight all subjects will be put on a calorie restricted diet (60% of baseline intake) to lose the weight. WHY? Because thermodynamics and energy balance.
I suggest you read and start your own blog if you feel this information needs to reach the public.
- again completely false - but I will leave it for You to work it out in Your spare time.
Please explain how the H+ from an NADH in the ETC that comes from the Krebs cycle from an acetyl-CoA that was derived originally from glucose differs from that of an H+ from the Krebs cycle from an acetyl-CoA that was derived originally from a fatty acid.
aaa aaaaa, we are talking KETONE BODIES!!, not fatty acids - please do not twist the story.
"….could enlighten me."
go PubMed
There is no medical solution. Losing weight and maintaining weight loss is hard. There is no magic macronutrient to gobble or shun that will cause guaranteed weight loss. There is no food that eliminated or increased in your diet will automatically shed pounds. Instead, it's CICO. It really is just that. You have to start by acknowledging that you eat too much. Then you have to determine through a series of experiments where the "too much" borderline exists and establish a whole new set of habits. Finally, you have to maintain those habits while living in a society which makes it all too easy to slip back into the old ways. It's hard. It's just freakin' hard.
James Krieger and Lyle Mcdonald have pointed to studies showing this is a neurological adaptation, not some kind of foregone biochemical conclusion like "dieting damages mitochondria".
I forget the details though ... how, specifically the muscle recruitment changes (or if muscle recruitment was even the mechanism of better efficiency) try searching their forums
The two Richard L. Veech studies are very interesting and they have abstracts! I don't see what either has to do with the orientation of Evelyn's post nor even with Christopher's wild inchoate comments in the thread although there is some delta G NAD etc awaiting, as we all are, his elucidation.
A quote from each abstract and a comment or two:
KETONE ESTER EFFECTS ON METABOLISM AND TRANSCRIPTION.
"... feeding ketone body esters leads to pure ketosis, unaccompanied by elevation of free fatty acids, producing a physiological state not previously seen in nature. "
"a physiological state not previously seen in nature." I like that. New territory. These ketone esters haven't made it into the metabolism textbooks yet, but they are a hot topic in LC circles. Ketone esters are the hottest thing since sliced bread! Errrr, make that the hottest thing since sugar alcohols. (haha, almost made a no no there!)
As an aside, referring to the discussion in this thread, ketone esters would be the exogenous ketones and the body could "tell the difference" between them and its naturally produced ketones, chemically identical though they be, by the FA concentration in the blood.
However, the "unaccompanied" by FA elevation part is very interesting as it explains a little of why ketones should be examined as part of the overall metabolism and not just as another pretty face or magic molecule ... and going now to Veech's next study which has more on the ketone FA connection.
THE THERAPEUTIC IMPLICATIONS OF KETONE BODIES: THE EFFECTS OF KETONE BODIES IN PATHOLOGICAL CONDITIONS: KETOSIS, KETOGENIC DIET, REDOX STATES, INSULIN RESISTANCE, AND MITOCHONDRIAL METABOLISM
"Current ketogenic diets are all characterized by elevations of free fatty acids, which may lead to metabolic inefficiency by activation of the PPAR system and its associated uncoupling mitochondrial uncoupling proteins. New diets comprised of ketone bodies themselves or their esters may obviate this present difficulty."
Current ketogenic diets go with elevated FA which may lead to metabolic inefficiency... UCP! I wonder how that fits in with ketone/FA efficiency.... hmmm, well, as Evelyn says above in her top post, a subject for another day.
I think Christopher is uncoupled from the reality that ketones or glucose don't feed into the ETC. Which is my point, and thank you all who got it! From acetyl-CoA forward the pathways are identical. It is also a stoichiometric process (whole number particle ratios, fixed ratios) up until the point of ATP synthase or decoupling. But the non-stoichiometric electrochemistry there is not magic either. For another day. :-)
On the internet, no one knows you're a dog, but in this case I really am telling the truth.
"He rolls his eyes and shakes his head at your argument." - I am suer he did, if he is a member of CICO club, like you are , he must be a one smart cookie.
I did not state any argument or make any comments - I wanted to see what kind of biochemists are you with such a simplistic, high school level, view of cell energetics posted on the Internet with Charles Darwin citation at the top.
"... you're a dog, but in this case I really am telling the truth."
you are most welcome to call me whatever you like, people whom I treat seem to thing otherwise, but it is possible that they also call me a dog behind my back, but than why they would come back for a treatment?
And that would be all.
What the ATP is actually going to be used for is determined by macronutrients, macronutrients, hormones, activity and so on.
Oxidative metabolism: glucose versus ketones.
Prince A1, Zhang Y, Croniger C, Puchowicz M.
Author information
Abstract
The coupling of upstream oxidative processes (glycolysis, beta-oxidation, CAC turnover) to mitochondrial oxidative phosphorylation (OXPHOS) under the driving conditions of energy demand by the cell results in the liberation of free energy as ATP. Perturbations in glycolytic CAC or OXPHOS can result in pathology or cell death. To better understand whole body energy expenditure during chronic ketosis, we used a diet-induced rat model of ketosis to determine if high-fat-carbohydrate-restricted "ketogenic" diet results in changes in total energy expenditure (TEE). Consistent with previous reports of increased energy expenditure in mice, we hypothesized that rats fed ketogenic diet for 3 weeks would result in increased resting energy expenditure due to alterations in metabolism associated with a "switch" in energy substrate from glucose to ketone bodies. The rationale isketone bodies are a more efficient fuel than glucose. Indirect calorimetric analysis revealed a moderate increase in VO2 and decreased VCO2 and heat with ketosis. These results suggest ketosis induces a moderate uncoupling state
I wish you all happy and satisfying lives.
Adv Exp Med Biol. 2013;789:323-8. doi: 10.1007/978-1-4614-7411-1_43.
Oxidative metabolism: glucose versus ketones.
Prince A1, Zhang Y, Croniger C, Puchowicz M.
That final line should read:
"These results suggest ketosis induces a moderate uncoupling state and less oxidative efficiency compared to glucose oxidation."
All in all, a more complete statement as the missing clause makes explicit that which was only implicit in the first part of the sentence.
~
Why are you here, Christopher? Have you read enough of this site to know what it's about? Just curious.
This study is interesting, a nice counterpoint to the two by Veetch. Keep them coming, and if you actually find you have something to say on metabolism, preferably without the drama, don't be shy.
yes you are 100% right it does not apply to human nutrition - it applies to energy balance from the point of the Complex I on ETC to the ATP generation and how H donors, FAD and NAD are influenced by glucose and ketones. You all seem to go wrrrrrrr, because I said that ketones might be good for us and, I gather because you think: this means: aa haaa ketones - low carb high fat ketogenic diet - this guy is LCHF !!!. I agree with your dietary choices - it was shown to be of great benefit to many, to say that carbohydrate rich diet is always bad is preposterous, the same as it is to say that ketones are to be ignored - I witnessed remarkable health improvements delivered by high ketones levels in blood (created by ketogenic diet).
Yes textbooks are expensive.
Sorry, I did not do it on purpose - I do not have hidden agendas. It is good that it happened however, because it tells me:
1) that you thing I am dumb to do such a thing without realizing that anybody can check it anytime
2) sorry to say but you do not understand this last statement and how it relates to the whole picture.
"Why are you here,"
I visited here not to change your world-view or dietary habits, which I condone, I was here because, well let me put it this way. If I was to take-on the world on the World Wide Web with "To kill an error…." Charles Darwin's quote at the top and followed by the post about cellular energy transduction, I would make damn suer that my understanding of the subject is more than mediocre high school level.
I wrote: "You truncated the last line of the abstract. On purpose?"
You truncated: "On purpose"
It's really too funny. My question marks are important. Look how your enumerated point one changes in meaning with my question mark in place! No, I don't think you are "dumb" but I do wonder how your error pointed out to you brings you to that conclusion. Do you feel "dumb"? And to the second point, why don't you come to the opposite conclusion, that indeed, I do have a clue to meaning of "These results suggest ketosis induces a moderate uncoupling state." Else, why do you think I looked it up? The statement in its truncated form is odd. And also, the truncation gives more important emphasis on your catnip toy, "uncouple." hmmmm.
It would be helpful, even a courtesy on your part, to tell us what you think and why you think it Articulate! Then we might be able to respond to you appropriately. As it is I feel like lecturing you on the importance of rat studies and the escalation to human studies when evidence is compelling. Does one need to point out to you that the scientists who write up these studies do not come to the exalted conclusions you seem to? Note in the quoted truncated sentence above, the word "suggests" is as far as it goes. It is the same in the other studies you quote, a perhaps here, a maybe there, and "suggests" in place of "it is conclusively proved."
You really should read some of the "in your face" topics on this site, you might find the Darwin quote is appropriate. You can read about carbsane in the pages of interest section in the right column, near the top of the page, the the flavors of the real thing, the topics themselves give a real savor though.
There is a lot of bad science propounded by self-appointed gurus or people seemingly in it for the money and this site is the only one that debunks them across the board, no matter who they are, nor what their credentials may be. Most internet debunking is of the straw man type or mere squabbles over territory or personality. Here you find the real thing (and some gossip, character bashing, fights without nerf bats, but hey, who cares and who's counting?). I had expected to spend the last decades of old age reading eighteenth century novels and tending garden, but delving into the fertile soil of metabolism, learning, debunking, and exposing pretentious gurus is a lot more fun.
I feel like I am trying to sell you something! So sorry. Let me hasten to add that there is something here to offend anyone and everyone. If you have a low tolerance for being in a state of cognitive dissonance you will find this place more of a purgatory than a paradise and it would be best for you to abandon hope and not enter here.
Do I ramble? Well too late now, but briefly, if you wish to debunk this site do it as Evelyn does, you owe it to her, with clearly stated purpose, facts (lots of them), science, and occasional wit. And it helps to be right too but not required. A well articulated argument goes a long way and everybody is wrong sometimes.
The work with exogenous ketones is very interesting and in trauma seems a worthwhile treatment to pursue. Which it is -- not unlike applying sugar straight to wounds and other applications like that. To translate this to the dietary context is also misguided.
There ARE no studies demonstrating net physiological benefits for truly ketogenic diets and no cultures who ever consumed such a diet for reference in a "real life" setting.
"That's frankly nonsense Billy."
I merely said to Christopher that he'd have a bit of an uphill battle here in gaining support for the net benefits of ketones, and as your response confirms the uphill battle he faces, it can't be said that my mentioning this fact was "nonsense." My comment was not some sort of slap-in-the-face to you; just that he shouldn't be disappointed in failing to get support here for his pro-position re the benefits of diet-induced endogenous ketones.
George Cahill, as you already know, was inclined to regard ketones a bit more favorably:
"...a chemical agent, β-OHB, that has played such a major role in man's survival may be expected to have actions other than simply calories. When nature has a beneficial substance, it may become pleiotropic through evolution with other survival advantages. Insulin and thyroid hormones are excellent hormonal examples...
β-OHBmay also have other metabolic effects than simply a fuel. Its role in diminishing muscle protein catabolism directly in fasting man and animals, is one example needing biochemical examination."
I wished to remind Christopher that your website is, after all, called CarbSane precisely because you long-ago rejected VLC ketogenic diets as being choiceworthy for health & well-being.
When did I do this? Context is everything here and this is simply not true.
In your quote from Cahill, he is describing a role for ketones that diminishes damage, not one that promotes health.
To prove the efficacy of their various passions, these partisans often haul out vaguely related but ultimately inapplicable "studies" on tiny, usually non-human populations of test subjects -- studies that have been published in obscure and inaccessible journals. So ketones -- well, ho-hum, more of the same.
Apologies,
I have indeed misread you.
Keto diets have their place. They have been demonstrated therapeutic in a VERY small number of conditions, and are being investigated in others.
Even in therapeutic contexts, keto is not always a miracle cure so there are tons of families out there who didn't see their child transformed into a normal thriving kidlet despite trying the diet.
Even VLC for weight loss has its applications, but there is no long term data for such diets in weight maintenance to look at especially this keto variety where protein is also restricted.
Exogenous ketones are a different story as well. You cannot extrapolate the metabolic consequences of injecting them or ingesting them directly to a ketogenic diet. You just can't.
Tell me about it!
Stella—in my wallet—right next to my Concealed Carry Permit is my Medicare card...
"The Glenmorangie buzz..."
Greta—two thumbs up for having the good sense to treat yourself with the high-class stuff:
"Glenmorangie...has been called delicate and mild, even faintly sweet; Glenfiddich, fruity and well-balanced; the Glenlivet, mellow, ripe and peaty; Macallan, my favourite, powerful and yet smooth." [Kingsley Amis. Everyday Drinking. (2008) p. 121.]
(Uncle Jack [Daniel's] on-the-rocks for yours truly.
You see Carbsane, what is the point to have a dialog with you, who has such a naive understanding of the matters at hand. Please find a physician who treats patients with restricted carbohydrate diets. Go for a coffee together, talk to her/him, talk to the treated patients - then we might had a dialog, wishing you a sunny day!
Well, I know exactly what I was doing when JF Kennedy was assasinated
'We observed a reduction in ROS [reactive oxygen species] levels after treatment with ketone bodies. Of note, the anticancerous property of several phytochemicals and dietary compounds is mediated by their antioxidant activity [48]. Recent studies indicate that β-hydroxybutyrate, the main ketone body found in the body, reduces oxidative stress [49] and, in turn, functions as a histone deacetylase inhibitor. Inhibition of histone deacetylase activity can suppress or prevent cancer growth as indicated by several studies with histone deacetylase inhibitors that are currently being evaluated for cancer prevention [50].'
well, I have a higher degree in pure mathematics - but the fact that one can add and subtract does not mean that one is not dumb. (I am not a mathematician - it is just a hobby).
However this is the Internet and I could be just a dog.
I managed not to truncate your text - hurray !!
I'll leave you to it good people.
Good by.
"You can't generalize that to the idea that a ketogenic diet is a healthy diet for everyone."
Stella—I think if you re-read my comments you will find that I did not
myself claim the above. Merely that ketones do have demonstrated physiological benefits that set them quite apart from the wheat-germ, the oat-bran, and the other purported "magic substance" cure-alls on your list.
I did not claim that ketones per se were a panacea for all the ills that flesh is heir to. I was merely going to bat for the poor beat-up ketone. But my sticking up for the ketone is not equivalent to my claiming that a VLC ketogenic diet is a "healthy diet for everyone."
Because while I think that there is good evidence [see the Cahill papers] of the physiological benefits of ketones—they do, after all, come to our rescue almost every day after a good night's sleep—I agree with our moderator that the deliberate choice of a ketogenic level of carb—and protein!—restriction is not only unnecessary for health & well-being but leads to abnormally elevated NEFA's [insulin falls so low it's unable to inhibit lipolysis]—not a good thing— and risks sarcopenia to boot. Not to mention the lesser but still unwelcome effects of insomnia, elevated cortisol, palpitations, etc.
"Nutty ketosis" is CarbSane's term—and while the ketones per se have benefits, the vigilant dietary restriction required to get them and maintain them is, I think, what she means by "nutty." For most folks, I agree with her that these various "costs" secondary to a VLC ketogenic diet are not offset by any "benefit" of elevated ketones.
I do not think, for example, that Peter D [Hyperlipid] is nutty. But his own VLC ketogenic diet strikes me as bizarre indeed—6 egg yolks fried in butter for breakfast, 100g Lindt 90% chocolate bar for lunch, some sort of very low-carb stew/chili for supper. Day in day out. I am unable to believe that such dietary restriction will buy him the healthy-longevity benefits he hopes for.
One of the expressions coined by our moderator that I regard as especially apt is "restriction addiction." I think this captures a most important feature of the whole business of the search for optimal diets. Nietzsche somewhere described the common practice wherein a
person becomes dissatisfied with his/her situation [weight, fitness, etc.] and resolves to start on a new path to correct matters. The
subsequent actions taken—whether new additions or restrictions—give an immediate boost to self-esteem because the agent has the sense of now being in control of things, taking charge, etc. Until, that is, sooner
or later when these same restrictions now come to feel increasingly oppressive, and eventually culminate in a new decision—to say, in
effect "F**k this!"—which gives a similar boost in self-esteem because the agent has the sense of once again "being the boss"—but now by breaking out of the very same self-imposed "prison" that provided the initial boost in self-esteem. Soon enough, this need
for feeling "in charge" returns, and the cycle gets repeated.
To me, this is a case of"Restriction addiction", and I think it's a chief explanatory factor for your list of various historical [and hysterical] purported magic dietary cure-alls—a list as lamentable as it is ludicrous.
"bleeding heart libertarian"☞ Like the lefties [but unlike Ms Rand]
I think [as the Ira Gershwin lyric goes] ♪♬'S wonderful,...'S marvelous♫ ♪ ♪—to help others less fortunate. Doing so, I
believe, is always splendid and praiseworthy.
But unlike the lefties, I think that the less fortunate nevertheless have no RIGHT to live at the expense of others— which would mean a compulsory obligation on the part of others to provide such assistance.
It's the compulsion that kills it—murdering the grace of giving.
Also like the lefties [but unlike Ms Rand] I have no fondness for the Captains of Industry—most of whom have no problem with legislation that restricts their competitors and enables them to remain
(as we used to say in Canada) Corporate Welfare Bums.
Or else (like a certain former resident of the White House) they were born on third base and grew up thinking they had hit a triple.
Perhaps the mathematics of statistics would be more practical as far as these studies go, but pure mathematics shows a refined nature, such a nature as balances your more confrontational posts giving a reason to check them out.
♪♬♩Dankeschön! ♫ ♪ ♪ [Greta—did you know that Bobby Darin had the rights to this song but gave it to Wayne Newton after hearing him at his first gig in NYC?—a wise choice, as it was Wayne's best...]
I admit it—my interests are all over the map.
The sidearms I favor are the Glock 21 (with Federal 230 grain Hydra-Shok JHP's) and the Sig Sauer P229 in .357 Sig caliber (basically a jacked-up 9mm round) that will deliver a Cor-Bon 115 grain JHP @ 1500 fps. Stopping power is not an issue with either pistol, though it is achieved by different strategies—the former via a big & heavy round, the latter via a much lighter bullet but at a greater velocity. The Sig 229 being the choice of the Secret Service, it's good enough for me! Both are reliable and capable sidearms.
Suitable, I should add, for, say, excursions off the beaten
path—but not something I find at all necessary for my usual adventures—a trip to Trader Joe's or Total Wine!
Post a Comment
Comment Moderation is ON ... I will NOT be routinely reviewing or publishing comments at this time..