Non-esterified fatty acid metabolism and postprandial lipaemia
Yet another gem from ... who else? ... Keith Frayn!
Non-esterified fatty acids (NEFA, or free fatty acids) are an important metabolic fuel. Both the concentration of NEFA and their flux through the circulation vary widely from hour to hour, reflecting nutritional state and physical activity. Inappropriately elevated plasma NEFA concentrations may have a number of adverse effects on both carbohydrate and lipid metabolism.
As my regular readers know well, this is a focus of my research.
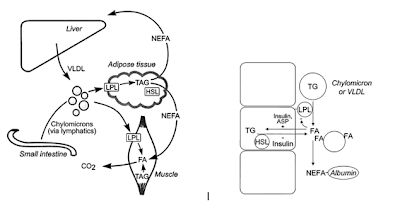
These adverse effects are likely to be most marked in the postprandial period, when NEFA release from adipose tissue is usually suppressed. Although the regulation of NEFA release in the postabsorptive state is well understood in molecular terms, the predominant pathway for release of NEFA in the postprandial state is the action of lipoprotein lipase (LPL) in adipose tissue capillaries on chylomicron-triacylglycerol (TG). Fatty acids released by LPL may either be sequestered in the adipocytes by esterification, or released as NEFA into the plasma. The regulation of this branch-point, which may be of crucial significance for postprandial metabolism, is not well understood. Factors stimulating tissue retention of fatty acids include insulin and acylation stimulating protein.
This summary is why studies like this one, that I've blogged on previously, do concern me. If you've achieved insulin-deficient nirvana through extreme carbohydrate restriction, you release NEFA in addition to those that escape from the adipose tissue. The NEFA levels nearly tripled four hours after the high fat low carb meal and appear to still be rising for the lean subjects, and they are significantly elevated above normal for the obese (almost 4X those of lean levels).
There is considerable indirect evidence that impaired regulation of this step occurs in insulin resistance and other conditions collectively recognised by an elevated concentration of apolipoprotein B (hyper-apo B). Inappropriate release of NEFA in the postprandial period is likely both to reduce the sensitivity of glucose metabolism to insulin and to accentuate postprandial lipaemia. Further study of the regulation of this pathway is much needed
Below is a side-by-side screen shot of NEFA for all tissues and Adipose Tissue metabolism:
Fatty acid movement across the adipose tissue capillary wall is bi-directional, unlike the situation in any other tissue (Fig. 3). In the fasting state, fatty acids are predominantly generated within the adipocyte by the action of HSL on stored TG, and flow in a net sense out into the capillaries and thus into the systemic circulation for delivery to other tissues such as skeletal muscle and liver. In the fed state, however, adipocyte fat stores must be replenished. This occurs by the action of lipoprotein lipase (LPL) in the adipose tissue capillaries upon circulating TG in the TG-rich lipoprotein particles, very-low-density lipoproteins (VLDL) and chylomicrons. In the postprandial period chylomicron-TG is the preferred and major substrate for adipose tissue LPL, competing with VLDL-TG to the extent that clearance of the latter in adipose tissue is markedly reduced [9–11]. The fatty acids released by LPL may flow into the adipocyte for esterification and storage as TG. Thus, the net transcapillary flux of fatty acids in the fed state must be inwards from capillaries to cell.
The implications of the emphasized statement above are at least something to be considered. Low carbers are usually very proud of their low fasting triglycerides. But what of 24-hour exposure when eating a high fat diet so that one's blood runs replete with chylos?
Even in normal, healthy subjects adipose tissue ‘capture’ of LPL-derived fatty acids never seems to be complete in the postprandial period. A proportion always ‘escape’ into the venous plasma. ... Thus, an important metabolic branch point may be identified in adipose tissue in the postprandial period. This is ‘downstream’ from LPL and determines the fate of fatty acids released by the action of LPL: entrapment in adipose tissue or release into the plasma. We and others have argued fully elsewhere [18–21] that efficient ‘trapping’ of fatty acids in adipose tissue in the postprandial period is a determinant of a ‘healthy’ lipoprotein phenotype, and that impaired trapping may lead to a multitude of adverse consequences, amongst which will be the generation of the features of hyper apo B [22] or the atherogenic lipoprotein phenotype.
As I've been trying to convey, putting fat where it belongs is a *good thing*. If we can't properly trap fat eating VLC, that's NOT.
The rate of fatty acid uptake and esterification is undoubtedly highly controlled. It has been known since early work with adipose tissue in vitro that this pathway is stimulated by insulin [34], and this is also clearly seen in vivo [35,36]. It has often been assumed that the action of insulin is to increase glucose uptake and hence the supply of glycerol 3-phosphate necessary for fatty acid esterification, but measurements in vivo suggest that the stimulation of fatty acid uptake by adipose tissue by insulin is not accompanied by increased glucose uptake: rather, an increased proportion of glucose is directed into glycerol 3-phosphate synthesis [36]. This implies that insulin stimulates directly either fatty acid transport or one or more of the enzymes of fatty acid esterification. Whilst insulin is undoubtedly important in the postprandial regulation of fatty acid movement in adipose tissue, in recent years another regulator of this pathway has been described, acylation stimulating protein (ASP) [37,38]. ASP is a more potent stimulus to fatty acid uptake and esterification in adipocytes than is insulin. ASP is the product of the interaction of components of the alternate complement pathway secreted by adipocytes themselves, and is identical to the peptide otherwise known as C3a-desarg [38]. Its production is markedly stimulated by the presence of chylomicrons [39]. Thus, ASP can be seen as a key part of a system of ‘microenvironmental regulation’ [40] in adipose tissue...
...In respect of glucose metabolism, elevated NEFA concentrations will impair glucose uptake in skeletal and potentially cardiac muscle by mechanisms which are part of the glucose-fatty acid cycle [43,44]. The net effect will be a diminution of sensitivity of glucose metabolism to insulin. This will be reinforced by the marked stimulatory effect of NEFA on hepatic glucose output, usually suppressed during the postprandial period [45]. In addition elevated NEFA concentrations may reduce hepatic insulin clearance [46], thus leading to systemic hyperinsulinaemia and subsequent downregulation of insulin-sensitive processes. Inappropriate NEFA release in the postprandial period can therefore be seen as a cause of insulin resistance and potentially risk of type 2 diabetes, and elevated NEFA concentrations are indeed a risk marker for that condition [2].
It's amazing the things one discovers following the work of researchers forward ... or in this case sideways. Please do go read this in its entirety for yourselves. I've probably not done it justice even with extended excerpts.
Comments
So, how much carb do you want us to eat, in what circadian pattern, and does the origin (e.g., chocolate fudge v. kale) matter?
Science-blogging is not when you make like Tycho. Science-blogging is when you make like Kepler!
here's a quick sampling of what's out there
The Jaminets (perfecthealthdiet.com) put it at 600 calories of glucose per day for mucus production (important to protect against H. Pylori infection & related putative cancer causing agents).
Lyle McDonald suggests if you are obese and losing steadily you don't need carbs more than once per 2 or 3 weeks and he suggests (this is from memory)- 2 big starchy meals (avoid fatty stuff that's easy to eat too many calories from for this) every 4 weeks plus occasional small cheats, ONLY if they help you stay on the diet.
The closer you get to 10% body fat the closer together your carbohydrates should be because at lower bodyfat the body makes more drastic metabolic adjustments.
Then there are Martin Berkhan, Alan Aragon and Anthony Colpo all of whom from what I'm reading suggest you don't radically cut carbs, just calories.
wrapping up with a repeat: it doesn't look like CarbSane is or will be providing specific diet advice or specific numbers
> tissue ‘capture’ of LPL-derived fatty acids
> never seems to be complete in the postprandial
> period. A proportion always ‘escape’ into
> the venous plasma
should provoke anxiety
No, good post because this is clearing up a lot of questions I've had about the overconsumption of sat fat that's pushed in the the LC community. Although I fell for it, I always wondered why I should follow a diet that's made up predominantly of what I was trying to lose! Even when I was at my most fanatical LC stage, I still questioned the macro breakdown ...
It's frustrating to keep finding all of this information that - while not settled science - goes counter to so many things the low carb community has been mis-educated with.
I read mostly science blogs, and it took me a long time to find you and other science-based nutrition bloggers, such as Drs Guyinet and Jaminets. Tagging via researchblogging.org gets your work out to lots of folks like me, as well as other scientists and science writers (often a lot of overlap there). Although I wish I would have found you sooner, I'm so glad I finally stumbled into your great lab/living room!
Thanks-
aek
Am I reading this right, or is the implication from the research you've quoted that following a VLC diet may actually increase the risk of type 2 diabetes due to inappropriate NEFA release?
So, yes, King, that is my tentative interpretation/concern. Just like glucose -> glycation -> problems, so too does NEFA -> problems (although the mechanisms are more varied by tissue apparently and less well understood). And while pp glucose spikes come down, the NEFA release seems to be more chronic even for pp-induced levels. This may be why the Mediterranean diet ultimately worked better for diabetics in Shai.
My perspective is that excess glucose and FFA are both damaging, particularly in combination. But what does "excess" mean? I think the key word in Frayn's sentence is "inappropriate" release of FFA.
I think you know about the Randle cycle. Glucose and FFA are in a sort of balance with one another. Postprandially in a lean healthy person, a carb-containing meal increases glucose and insulin, and FFA are suppressed by 80-95% for hours. Without carb, FFA may increase postprandially. But there's no glucose spike in that case.
Personally, I think the most dangerous thing is when you have someone who doesn't adequately suppress FFA during a glucose incursion. Then you have high levels of both. That seems to result from insulin resistance preventing the complete suppression of hormone-sensitive lipase. The insulin resistance is probably mostly the result of chronic cellular energy excess in my opinion, and that in turn is mostly due to energy imbalance. Obesity is a marker of energy imbalance but lean people can be out of balance as well (and that may be the most dangerous because adipose tissue isn't taking the pressure off the lean tissues).
The point is that in my opinion, elevated FFA without elevated glucose may not necessarily be "inappropriate". It occurs normally during fasting, for example. Yes, elevated FFA inhibits glucose metabolism and insulin's glucose-suppressing action, but it kind of makes sense from a physiological standpoint. I'm not convinced it's pathological. When you have high postprandial glucose but you fail to suppress FFA, like one sees diabetes and upper body obesity, then you're in trouble.
I totally agree with Stephan here. I've read the Frayn paper and most of your posts on NEFA. I have to say you are threatening to make the same mistake your friend GT is making - confusing internal metabolite behaviour with food behaviour
Hyperglycemia bad ergo eating carbs bad
vs
Inappropriately elevated NEFA bad ergo eating a lot of fat possibly dangerous
Both non-sequiters
Maybe focus more on the "hyper" and "inappropriate" modifiers.
The question is which foods (or non-foods) are causing what. if you have read any of my recent posts, I think "macronutrient ratios" is barking up the wrong tree as the concept of macronutrient itself is bereft.
It's about the poisons that cause metabolic syndrome, not mixing "macro" ratios like you are making a cake.... or fearing either of the two evolutionary fuel sources - starch or LCFAs.
Re: your personal "heart racing". VLC, to the degree it may create relative hypoglycemia, is likely to have periods of elevated epinephrine where your adrenal glands are trying to liberate glucose from your liver to drive up BG -this is just homeostasis, no matter how uncomfortable, and as long as you were in sinus rythym, would have zero relationship to the type of arrythmia that you seem to be worried about. It has nothing to do with an arrythmia precipitated by altered metabolism in someone with a fresh infarct du to fatty acids. People on VLC or atkins induction report tachycardia all the time. I don't recommend steady ketosis, but more arrythmias are associated with high carb than with low - esp atrial fibrillation, which is serious.
Could be dangerous - in the same way exercise would be -IF you had a pre-esisting conduction abnormality or valvular aortic stenosis or IHSS - but the solution is higher carb intake to avoid hypoglycemia, not lower fatty acid consumption
Have you even read the above quoted research? I realize it's challenging to read and understand (because it's very technical and littered with acronyms), but it really is a bit of an eyeopener.
And I highly disagree that the concept of macronutrients is bereft. As calories go, it's all energy to the body, but the source and quality of the foods you eat really does matter for your health.
Re: The point is that in my opinion, elevated FFA without elevated glucose may not necessarily be "inappropriate". It occurs normally during fasting, for example. Yes, elevated FFA inhibits glucose metabolism and insulin's glucose-suppressing action, but it kind of makes sense from a physiological standpoint. I'm not convinced it's pathological. When you have high postprandial glucose but you fail to suppress FFA, like one sees diabetes and upper body obesity, then you're in trouble.
I look at this from the POV of if low carbing is potentially dangerous. I can't change whatever risk I took with the way I lost my weight, but I can change what I do now.
NEFA is almost completely ignored in the low carb community, and actually celebrated (anything that increases lipolysis) even when there's no indication of actual increases in total oxidation rate. You have a lot of IR and T2's "self treating" and lowering BG levels. However, my research demonstrates in most, the "inappropriate" NEFA release from adipose tissue PREcedes the hyperglycemia. The study I linked to () demonstrates an inappropriate NEFA release in both lean and obese subjects. This study (http://www.ajcn.org/content/91/3/578.abstract- BTW if you have access to the full text I would love a copy!) showed increased fasting and 24hr exposure to FFA's. They correlated with increased LDL - something that is very common on low carb diets.
I've come across a virtual tsunami of literature and studies indicating that chronic IR follows and is instigated by elevated NEFA. If a dietary approach increases NEFA while decreasing a downline symptom (BG) it can potentially cause a progression of IRS and the related risks. That certain other biomarkers, like fasting triglycerides, are generally lower may (note the may) not be reflective of what's really going on and provide false assurances.
I've got several related posts in the works. I hope you'll read them and provide feedback.
Where BG's are concerned in the IR/T2, spikes do still come down in a matter of hours. But look at the pp NEFA's in the above-linked study. They persist 4 hours out and look to be on the uptick. Thus while pp hyperglycemia does not seem to lead to chronic hyperglycemia, pp hyper-NEFA can, apparently lead to a chronic state.
So you are not concerned when people report tachy on VLC??
As to: Just to be clear, tachycardia due to epinephrine in response to hypglycemia, not a result of high NEFA per se.
Could be dangerous - in the same way exercise would be -IF you had a pre-esisting conduction abnormality or valvular aortic stenosis or IHSS - but the solution is higher carb intake to avoid hypoglycemia, not lower fatty acid consumption
From what I've been reading, NEFA doesn't *cause* arrhythmia or ischemia, but it can interfere with recovering from it. So this can be a double whammy with hypo -> arrhythmia and NEFA -> failure to recover. Why take that chance?
As you say, the solution is to eat more carbs. YES!! But don't tell this to most low carbers who insist that VLC diets are the healthiest option for everyone.
I believe that the episodes from LC stint 2 that have been almost absent this go-round are due to my "cheats" (essentially carb-ups) and to my consumption of leaner proteins.
I think you're somewhat misinterpreting my position on fat intake. I don't fear fats, but do believe that the "moderate protein" (which ends up being rather low on many plans), "high fat" versions of VLC *MAY* be harmful. Just because certain fats aren't harmful in large amounts doesn't mean that eating large amounts is "healthy". Make sense? I do worry for those eating VLC who are either constantly regaining and losing (even the same 5 pounds but often more) having plateaued out at a higher than "normal" weight. I'm a very dense outlier in this regard, but I do still have fat left to shed and hence my personal concern.
In the end if you eat more carb you have to eat less fat or ... you'll get fat!
I do hope you'll read some of the stuff I'll be posting in the coming weeks.
I think excess circulating energy in any form, whether from FFA or glucose, is harmful. I certainly don't think FFA should be ignored, as they are potentially just as damaging as glucose in my opinion. My main point is that the concept of "excess" is poorly defined in the case of FFA. We know a fair bit about what excess FFA means in the context of a typical mixed American diet (or at least we have correlations), but not much about what it means in a low-carb context in which the metabolism resembles fasting in some ways.
Regarding the increased heart attack risk in morning, I think it's tough to pin that on FFA. You've just been laying still for hours, and you might dislodge a thrombus by getting up. There are many things that are physiologically different about the morning relative to the afternoon.
When you say elevated FFA precede hyperglycemia, are you talking about fasting hyperglycemia or postprandial hyperglycemia? That's a critical distinction, as the latter is a much more sensitive marker of glucose homeostasis problems. In any case, would you mind posting that reference?
I imagine you read the "gluttony and sloth" paper on lipotoxicity. They speculate that excess circulating FFA leads to ectopic lipid accumulation (muscle, pancreas, liver, etc.), which leads to glucose homeostasis problems. I think it's plausible. But what causes the excess FFA? Their model of choice for supporting their hypothesis is a db/db mouse, which has no leptin signaling and is therefore also profoundly insulin resistant. Insulin resistance in adipose tissue causes a defect in the suppression of lipolysis, the same way the liver doesn't suppress glucose production when it's insulin resistant.
And what causes that? Probably chronic energy imbalance, exacerbated by some other factors such as a lack of short-chain fatty acids, minerals, omega-3s, polyphenols, etc.
http://carbsanity.blogspot.com/2010/10/insulin-resistance-taubes-v-frayn.html
I just received the full text of Hernandez and will do a brief blog post on one aspect of it showing the NEFA levels throughout the day. They are elevated big time and consistently.
Re: NEFA and morning heart events, I don't think they are the instigators, but they might well be compounders.
I suppose there's enough content here and in my archives to start compiling more summary articles. I've got one on IR in the works. Certainly NEFA will deserve it's own treatment!
"Have you even read the above quoted research? I realize it's challenging to read and understand (because it's very technical and littered with acronyms), but it really is a bit of an eyeopener. "
Umm.. yeah, and whole lot more than that besides. I don't consider it challenging and this site (no offense CS) is hardly the discoverer of any of this info on free fatty acids. It's not really an eye-opener at all if you understand it. Only if you think eating food has to be dangerous!
What does THAT mean?
"the studies linked seem to indicate that a very low carb diet induces a pathological response"
That's your interpretation, it would not be mine. It is not pathologic.
" I've read (and enjoyed & agree with BTW) your macro-myth post and it's a good one. But in the end all dietary LCFA's must be "cleared" relatively rapidly, and failure to do so indicates metabolic dysregulation."
Thanks. Yes, but there is no evidence the LCFA is causing the metabolic dysregualtion. Even granting some of the bad effects of metsyn could be mediated via inappropriate elevation of NEFA, who would trade guaranteed hyperglycemia for the speculative effects of elevated NEFA. We've just agreed you have to eat something. The randle cycle guarantees that there will be an inverse relationship between glucose and ffa availability. The only way to avoid this is to be dead. You can't , a a la Dr. Davis, eat in a way that absolutely minimizes both. This reminds me of telling diabetics how important it is to "manage their cholesterol" while advising them to eat a low fat diet full of glucose.. We know that advice is a massive fail.
"Where BG's are concerned in the IR/T2, spikes do still come down in a matter of hours. But look at the pp NEFA's in the above-linked study. They persist 4 hours out and look to be on the uptick. Thus while pp hyperglycemia does not seem to lead to chronic hyperglycemia, pp hyper-NEFA can, apparently lead to a chronic state."
It's not leading to anything. If you eat to minimize the NEFA, you will maximize the heck out of your serum BG, because you cannot eat isocaloric without massively increasing carbs if you minimize LCFAs. Or you can do like Davis and substitute PUFA WHICH IS LIKELY CAUSING THE WHOLE METABOLIC DYSREGULATION IN THE FISRT PLACE.
OK, sorry to shout, but that is the point of my no such thing as a macronutrinet posts - the way to address the met dysreg is avoid hyperlgycemia (we know that is bad) and eliminate the things causing met dyreg - I suggest wheat fructose and linoliec acid. There is simply no evidence whatsoever that eating LCFAs causes met dysreg. Zero. But there is good evidence for avoiding the NAD (neolithic agents of disease)
"So you are not concerned when people report tachy on VLC??"
I am not concerned about sudden death in normal people- simus tachycardia would be an antidote to many deadly arrythmias, in fact. The NEFA related one you've written about is in damaged heart muscle.
"From what I've been reading, NEFA doesn't *cause* arrhythmia or ischemia, but it can interfere with recovering from it. So this can be a double whammy with hypo -> arrhythmia and NEFA -> failure to recover. Why take that chance?"
Life is full of chances and conditional probabilties. If by avoiding sat fat you increase your risk of atherosclerosis - ischemia, what consolation is it that you're having a heart attack but theoretically less likely to have an arrytthmia from it?
"As you say, the solution is to eat more carbs. YES!! But don't tell this to most low carbers who insist that VLC diets are the healthiest option for everyone. "
That is the solution to avoiding elevated epinephrine and tachy, but not becaase I think you will die from it and it is not NEFA that is causing it.
"I believe that the episodes from LC stint 2 that have been almost absent this go-round are due to my "cheats" (essentially carb-ups) and to my consumption of leaner proteins."
may well be the case, and I'm glad to hear that. I have less orthostasis on 20% carbs than 10%.
"Just because certain fats aren't harmful in large amounts doesn't mean that eating large amounts is "healthy"."
I agree in principle, but I have been trying to look for evidence that any dietary saturated fat (not pufa) can per se cause anything pathologic and I have found nothing. The cardiologists look desperately to prove it and cannot, but try to interpret their own findings that way anyway.
"Make sense? I do worry for those eating VLC who are either constantly regaining and losing "
Again, I worry that as Stephan has said, that it is counterproductive for several reasons to be VLC. But I think it is that you are losing the benefits of starch in the diet and that you are unnecesarily limiting yourself on VLC - not that the high level of fat itself is of any harm at all. It makes no sense to me that 25% of calories from sat fat is OK, but 30% is dangerous. It makes sense to me that 20% from starch is better than 10% though.
What does THAT mean?"
I mean it is not an eye- opener. It is not surprising. I am not shocked. This is not the first time I've heard of this. If you are convinced, like most of the population, that FATS somewhere, somehow, must be deadly, then you can read these papers and be alarmed. I am not alarmed, this is not new information.
If a person is searching for evidence of what dangers lurk in their diet, like, "hey did you know that there are FATs in your blood that go up on low carb? OH no!" then they might be alarmed. That is what I see happening here, as I've tried to make clear.
Clear enough?
To say something is an eye-opener means the person should be surprised or shocked or worried.
I am not shocked. It is not news to me and I don't interpret it the way you do.
When you say "my research" do you mean your reading of published papers, or your own actual research? Do you do research in this area?
And one of the things that concerns me here is how can you even quote something if you have only read the abstract? That is no better than a press release really. This is one of the ways it is easy to get confused, by giving any attention at all to a mere abstract.
How does one reduce NEFA? I am NOT advocating a low-fat lipid-hypothesis-reasoned approach. NEFA is largely controlled by adipose cell function and release from fat cells in normal people. This paper also discussed how impaired trapping can exacerbate the problem because dietary fat is actually not supposed to add to the free fatty acid pool - it is supposed to be transported as triglycerides and taken out of circulation following lipolysis. So, it is not the dietary fats that are causing anything, saturated, PUFA or otherwise. It's the disregulated fat cell that improperly releases the NEFA and/or fails to properly take in FFA's hydrolyzed from chylomicrons that's the problem.
And here, low carb eating does lead to a disruption of this process in obese especially, but also in normal lean subjects.
Elevated NEFA directly stimulate basal insulin levels but inhibit acute insulin response. They inhibit insulin's vasodilatory effects. They inhibit NO production in vascular endothelium. Cause increases in intramyocellular lipids that cause peripheral IR if they accumulate in a hypercaloric state such that intermediates in lipid metabolism (e.g. ceramides and diacylglycerols) become ROS. I could go on.
None of this is eye opening or worrisome to you? I'll be posting some data from the full text of Hernandez in a bit. We're not talking just a little elevated. We're talking consistent and considerable elevation during weight loss on VLC. No concern? As you'll see, these are not normal physiological substrate levels!
I don't do primary research any more, but I had done it for almost a decade and was published in peer review journals. I'm well aware of the limitations of abstracts and try to avoid them. The other study was a full text, however, and now I've got the full text of this one to work with.
I think we're in agreement on missing out on the benefits of starch with VLC diets. High fat doesn't cause the high NEFA, severely restricted carbs do. Thus some carb consumption can improve NEFA and this should actually (as Shai demonstrated) improve glycemic control.
T2's controlling their diabetes through VLC dieting are likely masking their hyperglycemia by keeping dietary carb low. But they're almost definitely exacerbating the NEFA. In the long term there are definite consequences attributed solely to high fatty acid concentrations that should not, IMO, be ignored. I'm not the only one saying this as I'm sure I've posted up quite a few reviews lamenting the lack of focus on this and perhaps overemphasis on just glycemic control.
Thanks for the discussion (and the explanation)!
"low carb eating does lead to a disruption of this process in obese especially, but also in normal lean subjects."
perhaps exacerbation, don't agree with "leads to" - not convinced it is worse than the alternative of hyperlgycemia if they have metsyn
"T2's controlling their diabetes through VLC dieting are likely masking their hyperglycemia by keeping dietary carb low."
Not masking. Not having it is not masking.
"In the long term there are definite consequences attributed solely to high fatty acid concentrations that should not, IMO, be ignored."
I don't agree that they are as definite or as dire as what we know are the consequences of hyperglycemia.
If I were as snarky as Peter I would say that that explains why Dr. Bernstein has killed so many type IIs with his diet : )
I had a friend with fasting BG of 185 who went down to 118 and lost 40 lbs on VLC and I am supposed to worry about his NEFAs?
There reason drug studies of glycemic control increase mortality is because they are doing it with DRUGs, including our friend insulin. I am aware of zero clinical studies that show increased death or morbidity due to elevated NEFA in diabetics on VLC. I would like to see them if you have them, maybe I just haven't seen them yet. OTOH we know how bad hyperglycemia is.
Thanks for the conversation. I need to go work on my "FH and lipid hypothesis post".
perhaps exacerbation, don't agree with "leads to" - not convinced it is worse than the alternative of hyperlgycemia if they have metsyn
1. Need not be either/or, and 2. NEFA plays a more central role in metsyn. Hyperglycemia is a downstream symptom. Read: http://journals.cambridge.org/action/displayFulltext?type=1&fid=804856&jid=PNS&volumeId=60&issueId=03&aid=804844&bodyId=&membershipNumber=&societyETOCSession=
Perhaps "masking hyperglycemia" was a poor choice of words. Masking their metabolic dysregulation by only looking at BG levels could definitely be an issue.
NEFA is NOT dietary fat (except in the case where dysfunctional body fat fail to take it up sufficiently). So we're not talking about the misguided notion of reducing cholesterol intake to lower cholesterol levels, or of LDL clogging arteries or any of that.
We're talking the direct, well documented but as yet not always well understood as to the mechanism of which, actions of free fatty acids on different cells in our bodies.
I am sorry but that just sounds crazy to me. The only possible way to unmask it to have higher BG - do you seriously think ANY effect of elevated NEFA can possibly outweigh blindness, amputations and kidney failure?
Or if you think ending up with kidneys that run on alternating current can be attributed to elevated NEFA in the blood, then we've already proved eating high carb to minimize NEFA is no solution at all, as 99% of type IIs are already eating to the ADA minumum of "150 g a day and sucrose is fine" - so as Dr. Phil would say, "and how is that working out for you"?
OTOH, I've seen type IIs become metabolically indistinguishable in every important way from normal people on VLC diets - losing all medication requirements along the way. Their A1C plummets and they lose weight and all clinical (that word again) signs of met syn disappear, including improvement in serious indicators of chronic renal disease.
But you think it might be better for them to take drugs or insulin and eat more glucose? Just to be safe?
Show me a SINGLE study that shows diabetics are clinically worse on any of these catastrophic end-points (MI, blindness, amputations, ED, claudication, infections, death, etc.) with dietary maneuvers that lower AUC for blood glucose.
You must be asserting this with regard to type IIs, as if you think that normal people are endangering themselves with peripheral muscle IR and higher NEFA than otherwise, it would be doubly true for those that have metsyn, but I've not seen a shred of evidence for that.
I must quote Mr. Hitchens here and say: "What can be asserted without evidence can be rejected without evidence." I just don't see the clinical evidence (these papers are physiology and speculation, not clinical science showing difference in outcomes) that might even begin to match the clinical evidence of the harm of hyperlgycemia
You seem very sharp and appear to be comfortable interpreting bench research, but some clinical perspective might make you interpret these studies with much more care and skepticism.
Like Stephan, I am willing to stipulate that NORMAL people should not strive to be on a ketogenic diet long term, for a variety of reasons. But none of my reasons would relate to thinking that LC diets can cause or in any way exacerbate the major known clinical end points we see in metsyn, even if fat metabolism stays abnormal once BG is normalized.
The consequences of hyperglycemia are simply devastating, and can be somewhat invisible to those who lack a great deal of clinical experience.
It's time to filter these papers through our "plausometers" and then try to square them with clinical observations. IMO, they don't look threatening at all when you have the big picture.
He, like Gary Taubes, speaks like a real genuine scientist. They BOTH are very intelligent individuals, as well as Urgelt of YouTube . They ALL understand claories are only one factor among dozens upon dozens in the etiology of obesity.
The chemical behavior of fat cell receptors is NOT understood. It is more than time you acknowledge that, CarbSane - as well as Krieger
Many, MANY, long term low carbers report issues controlling blood pressure, bad cramping, palpitations, pre-diabetes, insulin resistance, etc. There are a lot of LCers who do not lose weight with this approach.
I must say if I came to you as my doctor with my concerns and you brushed them off as there being a risk in anything, I would find another doctor. Because if my diet is doing something that is a known predictive risk factor for sudden cardiac death, don't you think that should be a concern?
The Shai study - clinical observation - demonstrates that the Mediterranean diet (the highest carb diet of the three interventions studied) produced the best results after 2 years for diabetics: http://carbsanity.blogspot.com/2010/09/shai-and-diabetes.html
Dr. Dansinger from Tufts "reverses diabetes" using a moderate carb low sat fat approach. Agree or disagree with it, he reports excellent clinical results in those who adhere to his approach.
If one controls the NEFA, they'll control the hyperglycemia. It is ultimately hepatic insulin resistance and not dietary carb in moderation that is the issue here. http://bja.oxfordjournals.org/content/85/1/69.full
When you speak of MetS you seem to be just looking at some of those markers if you say VLC-cured diabetics are indistinguishable from "normal". NEFA is a diagnostic marker of MetS.
There are deleterious effects attributable to the NEFA that are often attributed hyperglycemia because it is the most manifest diagnostic in diabetics. I have a post sometime today discussing hypertension.
Lastly, your tone leads me to believe you think I'm some anti-LC lipophobe. I'm not. I ate lower fat higher protein than most for weight loss because it works/worked for me. You seem to be confusing NEFA with dietary fat. Eating fat doesn't elevate NEFA. Eating too few carbs does seem to, especially if one is overweight/obese.
Insult me as a physician all you want, I am not "brushing aside" anything. I am saying you are confused and really have no idea what you are talking about at this point. Just like the many confused patients I've had that are convinced of all kinds of nonsense based on reading stuff on the internet.
"blood pressure"
Huh? VLC causes LOW blood pressure! I have never seen anyone have their BP go up on VLC, ever. You have a paper showing with a randomized trial that shows LC raises BP relative to HC?
"bad cramping"
This is NEFA mediated?
"palpitations"
Epinephrine release due to LOW blood sugar - that's LOW - it is usually temporary and has ZERO to do with metabolic syndrome - it is desirable to lower serum BG if you have metsyn, no?
"pre-diabetes, insulin resistance, etc"
peripheral IR is not per se pathologic. You are deeply confused about this and confusing others about it as well. Explain how low carb intake if you are pre-diabetic can make diabetes worse - nonsense. You can overeat CHO and drive up muscle IS as an adaptation, but this tells you nothing about what's happening to your liver. You and Matt Stone are similarly confused on this and what it means.
"NEFA is a diagnostic marker of MetS"
a MARKER is not a clinical event - you are confused about this. lipoproteins are markers. Hypertension is a clinical event.
Show me evidence of clinical endpoints made worse with VLC in diabetes.
You can't because it is all conjecture.
Hypertension, stroke, MI, death -these are clinical endpoints.
One last time - the idea that significant endpoints are mediated by NEFA is a hypothesis that for particular endpoints may or may not be true.
But you cannot claim that elevated NEFA is itself a clinical endpoint, as it is not. It is something you claim is mediating CLINICAL ENDPOINTs.
I am afraid you remain confused and are confusing others as well, which is why I and Stephan comment here.
PS I've read that one already.
Thank you both.
Glad you've enjoyed it. I am firmly into head beating against wall territory.
I gave it a shot. Good luck to the rest of you. Best of luck to you too, CS.
Post a Comment
Comment Moderation is ON ... I will NOT be routinely reviewing or publishing comments at this time..